- What makes silicon, and other semiconductors, special is that it is possible to promote one of the electrons to an empty board - the conduction band - where they can move freely. It's a bit like the 3-dimensional chess played by the point-eared Dr Spock in Star Trek.
- Find the conductivity of intrinsic silicon at 300 K. It is given that n i at 300 K in silicon is 1.5 × 10 16 /m 3 and the mobilities of electrons and holes in silicon are 0.13 m 2 /V-s and 0.05 m 2 /V-s, respectively (Set-2–May 2003) Sol: Intrinsic concentration (n i) = 1.5 × 10 16 /m 3. Mobility of electrons.
Charge carrier density, also known as carrier concentration, denotes the number of charge carriers in per volume. In SI units, it is measured in m−3. As with any density, in principle it can depend on position. However, usually carrier concentration is given as a single number, and represents the average carrier density over the whole material.
Cocktail maker app. Charge carrier densities involve equations concerning the electrical conductivity and related phenomena like the thermal conductivity.
For silicon, N cb doubles for about an 8 degree rise in temperature. Because of the larger band gap, there will be fewer conduction electrons in silicon than germanium for any given temperature. Index Semiconductor concepts Semiconductors for electronics Reference Simpson Sec 4.7. Atomic Number of Silicon. Atomic Number of Silicon is 14. Chemical symbol for Silicon is Si. Number of protons in Silicon is 14. Atomic weight of Silicon is 28.085 u or g/mol. Melting point of Silicon is 1410 °C and its the boiling point is 2355 °C.
Calculation[edit]
The carrier density is usually obtained theoretically by integrating the density of states over the energy range of charge carriers in the material (e.g. integrating over the conduction band for electrons, integrating over the valence band for holes).
If the total number of charge carriers is known, the carrier density can be found by simply dividing by the volume. To show this mathematically, charge carrier density is a particle density, so integrating it over a volume gives the number of charge carriers in that volume
.
where
- is the position-dependent charge carrier density.
If the density does not depend on position and is instead equal to a constant this equation simplifies to
.
Semiconductors[edit]
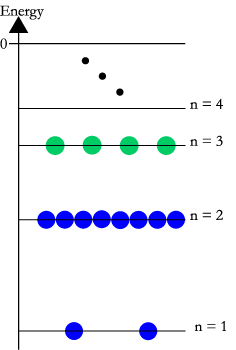
The carrier density is important for semiconductors, where it is an important quantity for the process of chemical doping. Using band theory, the electron density, is number of electrons per unit volume in the conduction band. For holes, is the number of holes per unit volume in the valence band. To calculate this number for electrons, we start with the idea that the total density of conduction-band electrons, , is just adding up the conduction electron density across the different energies in the band, from the bottom of the band to the top of the band .
Because electrons are fermions, the density of conduction electrons at any particular energy, is the product of the density of states, or how many conducting states are possible, with the Fermi–Dirac distribution, which tells us the portion of those states which will actually have electrons in ″them″
In order to simplify the calculation, instead of treating the electrons as fermions, according to the Fermi–Dirac distribution, we instead treat them as a classical non-interacting gas, which is given by the Maxwell–Boltzmann distribution. This approximation has negligible effects when the magnitude , which is true for semiconductors near room temperature. This approximation is invalid at very low temperatures or an extremely small band-gap.
The three-dimensional density of states is:
After combination and simplification, these expressions lead to:
A similar expression can be derived for holes. The carrier concentration can be calculated by treating electrons moving back and forth across the bandgap just like the equilibrium of a reversible reaction from chemistry, leading to an electronic mass action law. Atomic mass of n2. The mass action law defines a quantity called the intrinsic carrier concentration, which for undoped materials:
The following table lists a few values of the intrinsic carrier concentration for intrinsic semiconductors.
| Material | Carrier density (1/cm³) at 300K |
|---|---|
| Silicon[1] | 9.65×109 |
| Germanium[2] | 2.33×1013 |
| Gallium Arsenide[3] | 2.1×106 |
These carrier concentrations will change if these materials are doped. For example, doping pure silicon with a small amount of phosphorus will increase the carrier density of electrons, n. Then, since n > p, the doped silicon will be a n-type extrinsic semiconductor. Doping pure silicon with a small amount of boron will increase the carrier density of holes, so then p > n, and it will be a p-type extrinsic semiconductor.
Metals[edit]
The carrier density is also applicable to metals, where it can be calculated from the simple Drude model. In this case, the carrier density (in this context, also called the free electron density) can be calculated by:[4]
Number Of Electrons In Silicon-29
Where is the Avogadro constant, Z is the number of valence electrons, is the density of the material, and is the atomic mass.
Measurement[edit]

The density of charge carriers can be determined in many cases using the Hall effect,[5] the voltage of which depends inversely on the carrier density.
References[edit]
- ^Pietro P. Altermatt, Andreas Schenk, Frank Geelhaar,Gernot Heiser (2003). 'Reassessment of the intrinsic carrier density in crystalline silicon in view of band-gap narrowing'. Journal of Applied Physics. 93 (3): 1598. doi:10.1063/1.1529297.CS1 maint: multiple names: authors list (link)
- ^O. Madelung, U. Rössler, M. Schulz (2002). 'Germanium (Ge), intrinsic carrier concentration'. Group IV Elements, IV-IV and III-V Compounds. Part b - Electronic, Transport, Optical and Other Properties. Landolt-Börnstein - Group III Condensed Matter. pp. 1–3. doi:10.1007/10832182_503. ISBN978-3-540-42876-3.CS1 maint: multiple names: authors list (link)
- ^Rössler, U. (2002). 'Gallium arsenide (GaAs), intrinsic carrier concentration, electrical and thermal conductivity'. Group IV Elements, IV-IV and III-V Compounds. Part b - Electronic, Transport, Optical and Other Properties. Landolt-Börnstein - Group III Condensed Matter. pp. 1–8. doi:10.1007/10832182_196. ISBN978-3-540-42876-3.
- ^Ashcroft, Mermin. Solid State Physics. p. 4.
- ^Edwin Hall (1879). 'On a New Action of the Magnet on Electric Currents'. American Journal of Mathematics. 2 (3): 287–92. doi:10.2307/2369245. JSTOR2369245. Archived from the original on 27 July 2011. Retrieved 28 February 2008.CS1 maint: discouraged parameter (link)
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Charge_carrier_density&oldid=997871515'
Atomic Number of Silicon is 14.
Chemical symbol for Silicon is Si. Number of protons in Silicon is 14. Atomic weight of Silicon is 28.085 u or g/mol. Melting point of Silicon is 1410 °C and its the boiling point is 2355 °C.
Number Of Core Electrons For Al
About Silicon
Silicon is a typical example of metalloid, or the substance which looks like metal but does not have its properties. Its name came from the Latin word for flint, and in its pure form it looks like dark-blue metal. There is abundance of this chemical element on our planet, i.e. we can see it in the form of sand on beaches, or in the forms of various silicates. Silicon is considered to be one of the most useful metals for humanity since its alloys and chemical compounds have a large variety of useful properties. They are used, for example, for producing engines, transformers, various tools, popes, elements of roofs and windows, etc. Pure silicon is used for producing glass. As a very good semi-conductor, silicon is used for producing microelectronics, especially computers, laptops, etc.
Properties of Silicon Element
| Atomic Number (Z) | 14 |
|---|---|
| Atomic Symbol | Si |
| Group | 14 |
| Period | 3 |
| Atomic Weight | 28.085 u |
| Density | 2.3296 g/cm3 |
| Melting Point (K) | 1687 K |
| Melting Point (℃) | 1410 °C |
| Boiling Point (K) | 3538 K |
| Boiling Point (℃) | 2355 °C |
| Heat Capacity | 0.705 J/g · K |
| Abundance | 282000 mg/kg |
| State at STP | Solid |
| Occurrence | Primordial |
| Description | Metalloid |
| Electronegativity (Pauling) χ | 1.9 |
| Ionization Energy (eV) | 8.15169 |
| Atomic Radius | 110pm |
| Covalent Radius | 111pm |
| Van der Waals Radius | 210 |
| Valence Electrons | 4 |
| Year of Discovery | 1824 |
| Discoverer | Berzelius |
What is the Boiling Point of Silicon?
Silicon boiling point is 2355 °C. Boiling point of Silicon in Kelvin is 3538 K.
What is the Melting Point of Silicon?
Silicon melting point is 1410 °C. Melting point of Silicon in Kelvin is 1687 K.
How Abundant is Silicon?
Abundant value of Silicon is 282000 mg/kg.
What is the State of Silicon at Standard Temperature and Pressure (STP)?
State of Silicon is Solid at standard temperature and pressure at 0℃ and one atmosphere pressure.
When was Silicon Discovered?
Silicon was discovered in 1824.